To continue to rely on animal data as a means of protecting human health and the environment is tantamount to criminal negligence in the light of current science. The available technology is still a work in progress, but it is evidence-based and far more relevant to people than animal data
The EU chemicals program (REACH) was hailed as the most complex legislation in the European Union's history and the most important in 20 years when it was launched on 1st June 2007. It intended to "improve the protection of human health and environment through a better knowledge and risk management of chemical substances by industry".
Indeed, the European Chemicals Agency continues to receive accolades from industry and from the European Commission for the implementation of REACH, whose final registration deadline was 31 May 2018. Now is an appropriate time to take a deep breath and to examine the achievements, missed opportunities and any failings of the EU chemicals program.
Profit before planet
In terms of raw numbers, REACH has assessed little over half of its intended target of 30,000 chemicals over the course of its 11 year program. This is perhaps not completely surprising given that each chemical may need to undergo up to 11 different toxicity tests in order to determine its level of hazard to health and to the environment.
As an example, one of the toxicity tests - the rodent lifetime bioassay for cancer - requires rats to be observed for a period of two years. The aim of this article is not to criticise the 600 people at the European Chemicals Agency diligently working for REACH. Rather, the aim is to highlight the cost - in terms of animal suffering and human health - of ignoring current science and of an economic paradigm that puts profit above the future of this planet.
The EU chemicals program is clearly at odds with the precautionary principle. It states that even if a substance presents a risk to human health or the environment, authorisation may be granted if the socio-economic benefits are proven to outweigh risks arising from its use and if there are no suitable alternatives.
The synthetic chemical bisphenol A is emblematic of the most serious failings of REACH. Its use as a starting material for the synthesis of plastics is widely known. Recognised as far back as 1936 as being an oestrogen mimic, BPA’s endocrine disrupting effects on wildlife and humans began to appear in the early 1990s.
The European Chemicals Agency finally recognised BPA as an endocrine disrupting chemical in 2017, and a "substance of very high concern" (SVHC). To date, the European Chemicals Agency recognises 181 SVHCs out of the several thousand chemicals that it has assessed so far. If this tiny figure appears too good to be true, that’s because it is.
Limitations of testing
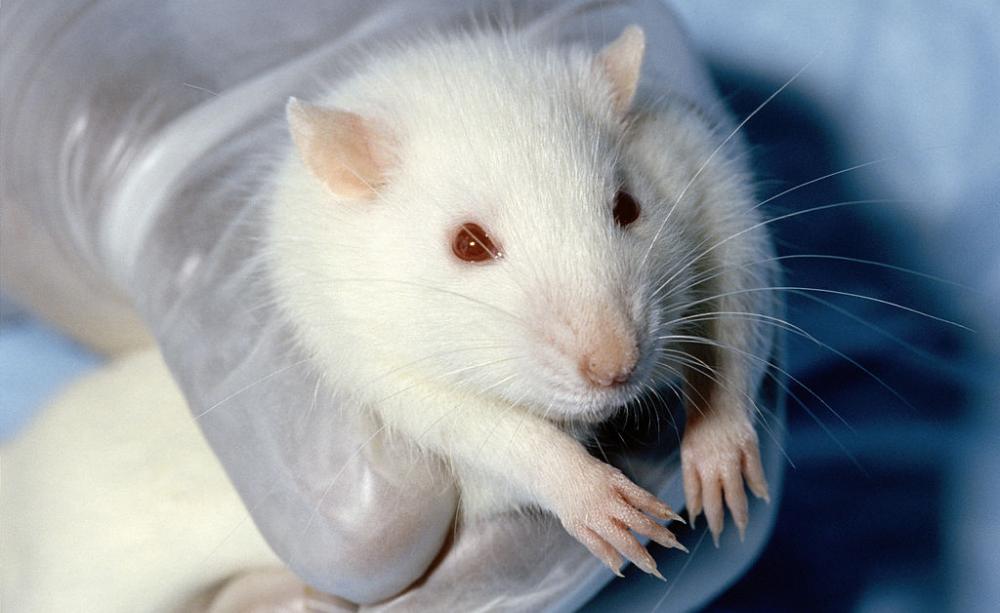
The traditional way to assess chemical toxicity is through animal experiments. This concept dates back to the era of the Nuremberg trials at the end of the Second World War. Our scientific knowledge has advanced since then in leaps and bounds, in particular the discovery of the human genome. Despite this, however, the need to provide animal data is still a legal requirement, which is good news for industry but bad news for everyone else.
To continue to rely on animal data as a means of protecting human health and the environment is tantamount to criminal negligence in the light of current science. The available technology is still a work in progress, but it is evidence-based and far more relevant to people than animal data
Not only is animal data much easier and faster to obtain than human data, but regulatory authorities trust industry to select an appropriate species of animal in order to assess its hazardous properties. In the example of BPA, the chemicals industry chose the Sprague Dawley rat to demonstrate the innocuity of this substance. This particular strain of rat is highly resistant to the hormonal effects of BPA.
Had the regulatory authorities insisted that the CF1 strain of mouse be used instead of the rat, the results would have been very different as the mouse is several thousand times more sensitive to the hormonal effects of BPA than the rat. Are humans more like mice or rats when it comes to BPA? The answer is that animals cannot predict human response.
Unlike pharmaceutical drugs, there is no legal requirement for industrial chemicals to undergo "post marketing surveillance" (aka pharmacovigilance). When severe adverse drug reactions come to light, health authorities can withdraw a prescription drug from the market rapidly. However, with industrial chemicals there may be little or no human data available, which equates to looking for a needle in a haystack to try to establish a cause and effect relationship.
The most reliable way to assess the effect of chemicals on human health is through the observation of human populations (epidemiology and biomonitoring) coupled with clinical and laboratory study of relevant human material - for example, analysis of blood and urine samples and the use of human cell cultures.
The way forward
REACH largely ignores human data in favour of animal data obtained through laboratory experiments. The ultimate responsibility for this state of affairs rests with the EU, whose agency, the European Chemicals Agency, oversees the implementation of this chemical program.
In the example of BPA, had the EU heeded the alarm bells of the 1990s, our health authorities would have had decades’ worth of stored human data - for example in the form of biomarkers - on which to base public health policy with respect to removing dangerous chemicals - a biomarker is a naturally occurring molecule, gene, or characteristic by which a particular pathological process or disease, can be identified.
To continue to rely on animal data as a means of protecting human health and the environment is tantamount to criminal negligence in the light of current science. The available technology is still a work in progress, but it is evidence-based and far more relevant to people than animal data.
"People, not chemicals, have the right to be presumed innocent until proven guilty. People also have the right not to be experimented on without informed consent; no one has ever been given the opportunity to grant or deny their consent before being exposed to the [toxic] burden that now contaminates us all" - J Thornton. Pandora’s Poison: Chlorine, Health and a New Environmental Strategy.
This Author
Andre Menache holds degrees in zoology and veterinary medicine and is currently scientific advisor to several NGOs dealing with public health and environmental issues.